Our Products
Showing(All)
Explore Our Products
Discover a wide range of high-quality pharmaceutical solutions designed to meet diverse healthcare needs.
-
Quick View
DIN INJECTION
Each 2 ml contains :
Drotaverine Hydrochloride I.P. 40 mg
Absolute Alcohol I.P. 8.0% v/v
Water for Injection I.P. q.s.
-
Quick View
LAXIL 100
Each 5 ml. contains:
Lactulose Concentrate USP
eq. to lactulose 3.35 gm
in a palatable base : q.s.
-
Quick View
MAPDIONE INJECTION
Each 0.5 ml contains:
Menadione Sodium Bisulphite 1 mg
Equivalent to Menadione 0.52 mg
Water for Injections IP qs
-
Quick View
Dol B6 Tablets
Each enteric-coated tablet contains:
Doxylamine Succinate USP 10 mg
Pyridoxine Hydrochloride IP 10 mg
Folic acid IP 2.5 mg
-
Quick View
CB 12 Injection
Each 1.5 ml (Part I) contains:
Vitamin C IP 150 mg
Each 1 ml (Part II) contains:
Vitamin B12 IP 2500 mcg
Folic Acid IP 0.7 mg
Niacinamide IP 12 mg
-
Quick View
Miracal D3 Syrup
Each ml contains :
Cholecalciferol IP
(Vitamin D3 IP) 400 IU
In a flavoured syrupy base
-
Quick View
Mapratone Syrup
Each 5 ml contains:
Thiamine Hydrochloride IP 2 mg
Riboflavin Sodium Phosphate IP 2.5 mg
Pyridoxine Hydrochloride IP 0.75 mg
Niacinamide IP 15 mg
D-Panthenol IP 3 mg
Cyanocobalamin IP 2 mcg
Flavoured Syrup base q.s.
-
Quick View
-
Quick View
Zincup Syrup 100 ml
Each 5 ml contains :
Zinc Gluconate USP
equivalent to
elemental Zinc 20 mg
in a flavoured syrupy base q.s.
-
Quick View
Zincup Syrup 50 ml
Each 5 ml contains :
Zinc Gluconate USP
equivalent to
elemental Zinc 20 mg
in a flavoured syrupy base q.s.
-
Quick View
Miracal Suspension 200
Each 5 ml of the Suspension contains:
625 mg Calcium Carbonate IP from
an organic source (Oyster Shell)
equivalent to
Elemental Calcium 250 mg
Vitamin D3 IP 125 IU
Flavoured Syrupy Base q.s.
-
Quick View
Miracal Suspension 100
Each 5 ml of the Suspension contains:
625 mg Calcium Carbonate IP from
an organic source (Oyster Shell)
equivalent to
Elemental Calcium 250 mg
Vitamin D3 IP 125 IU
Flavoured Syrupy Base q.s.
-
Quick View
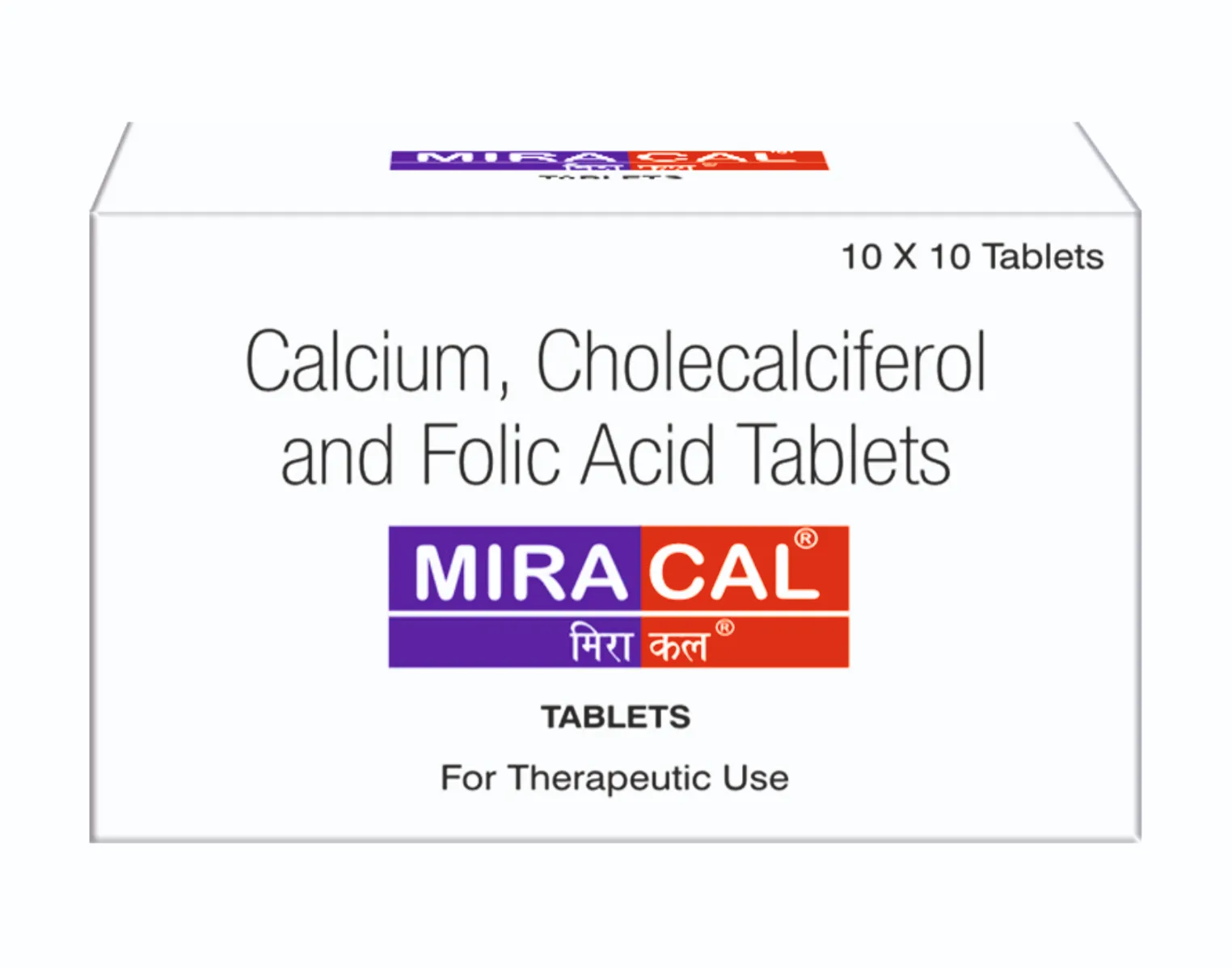
Miracal Tablets
Each uncoated tablet contains:
Calcium (as Calcium Citrate Malate) 250 mg.
Cholecalciferol IP 100 I.U.
(as Cholecalciferol Concentrate)
Folic Acid IP 50 mcg
-
Quick View
Fe-z XT Drops
Each ml. contains:
Ferrous Ascorbate IP
equivalent to Elemental lron 10 mg
Folic Acid IP 100 mcg
-
Quick View
Fe-z XT INJECTION
Each ml contains: Ferric Hydroxide in
complex with Sucrose equivalent to
Elemental Iron 20 mg
Osmolarity : Not less than 1150 and
not more than 1350 mOsmol/L.
-
Quick View
Fe-z XT Suspension
Each 5 ml contains:
Ferrous Ascorbate IP
equivalent to
Elemental lron 30 mg.
Folic Acid IP 550 mcg.
Flavoured syrupy base q.s.
-
Quick View
Fe-z XT Tablets
Each film coated tablet contains:
Ferrous Ascorbate IP
equivalent to elemental lron 100 mg Folic Acid IP 1.5 mg
Zinc Sulphate IP
equivalent to elemental Zinc 22.5 mg
Excipients q.s.
Colour : Red Oxide of Iron & Titanium Dioxide IP
Appropriate overages of vitamin added
-
Quick View
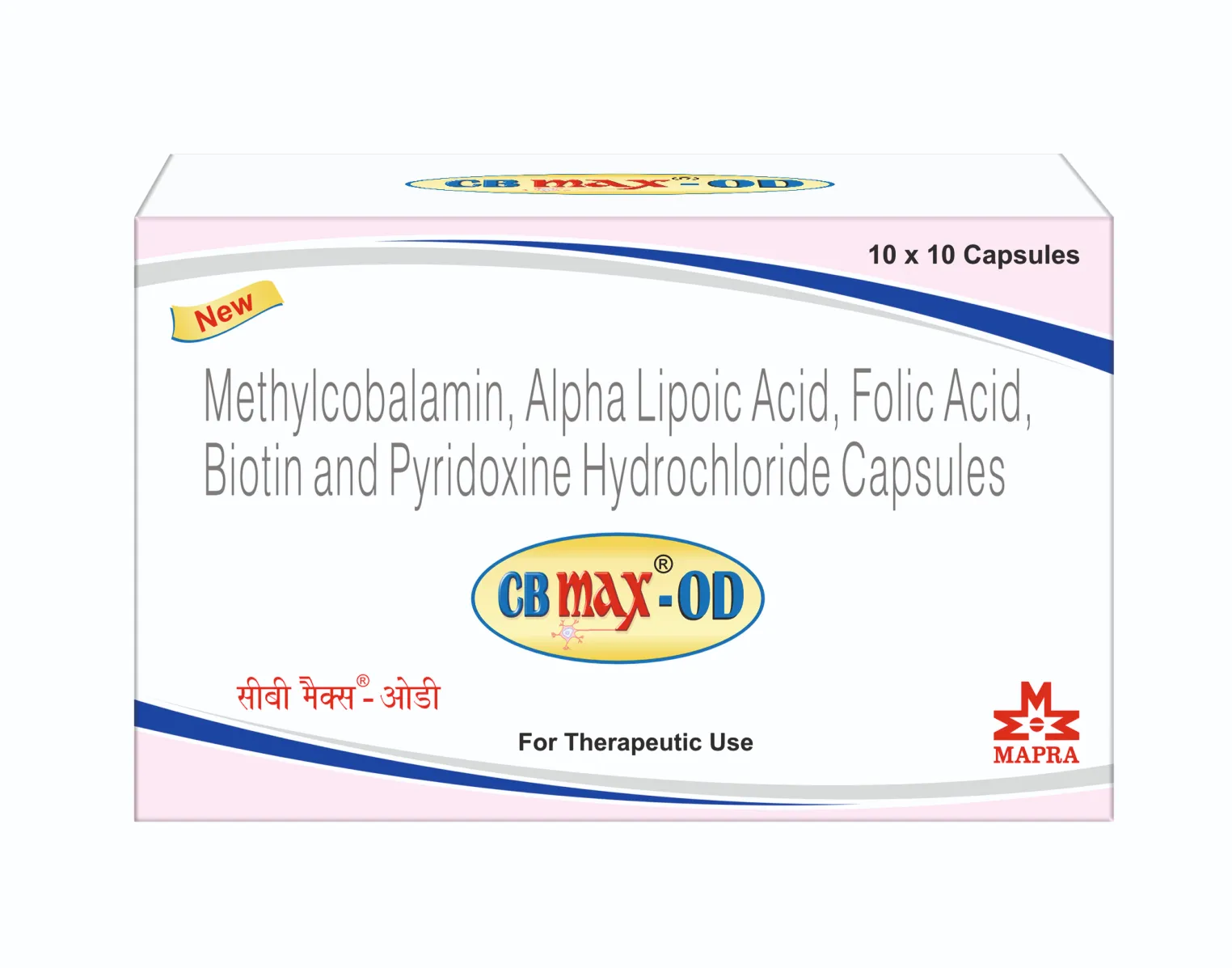
CB MAX -OD Capsules
Each hard gelatin capsule contains:
Methylcobalamin IP 1500 mcg
Alpha Lipoic Acid IP 200 mg
Folic Acid IP 1.5 mg
Biotin IP 30 mcg
Pyridoxine Hydrochloride IP 3.0 mg
(All in pellets form)
Colour used in product : Yellow oxide of Iron, Ponceau 4R & Titanium Dioxide IP
-
Quick View
CB MAX INJECTIONS
Mecobalamin IP 750 mcg
Pyridoxine
Hydrochloride IP 50 mg
Folic Acid IP 0.35 mg
Niacinamide IP 6.0 mg
Benzyl Alcohol IP 2% v/v
(As Preservative)
Water for Injections IP q.s.
-
Quick View
-
Quick View
-
Quick View
-
Quick View
Kofgard-LS Drops
Each ml contains:
Levosalbutamol Sulphate IP
Eq. to Levosalbutamol 0.25 mg
Ambroxol
Hydrochloride IP 7.5 mg
Guaiphenesin IP 12.5 mg
Flavoured Syrupy Base q.s.
-
Quick View
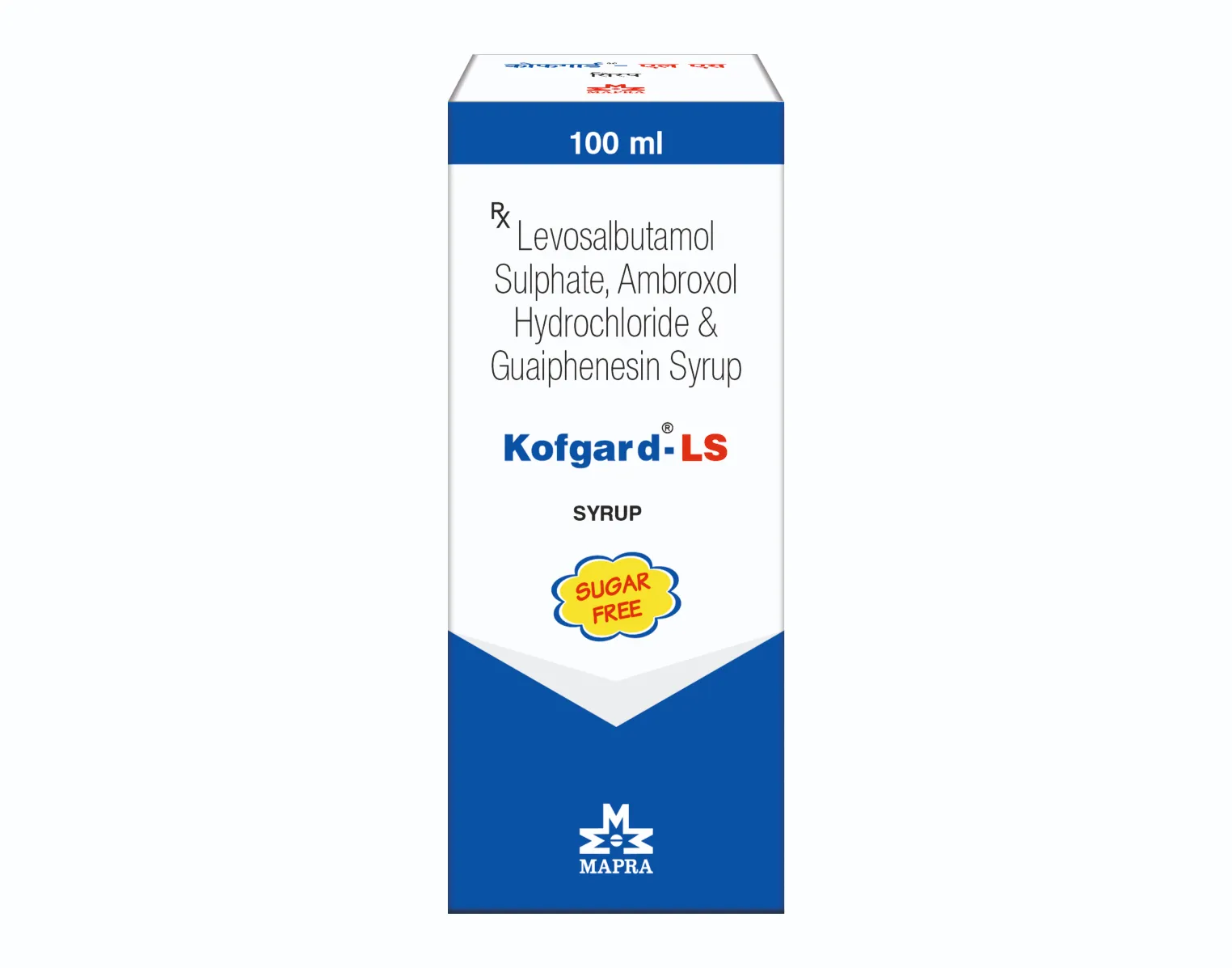
Kofgard-LS Syrup
Each 5 ml contains:
Levosalbutamol Sulphate IP
eq. to Levosalbutamol 1 mg
Ambroxol Hydrochloride IP 30 mg
Guaiphenesin IP 50 mg
Mentholated
Flavoured syrupy base q.s.
-
Quick View
Kofgard-D Syrup
Each 5 ml contains:
Dextromethorphan Hydrobromide IP 10 mg
Chlorpheniramine Maleate IP 2 mg
Mentholated
Flavoured syrupy base q.s.
-
Quick View
Monet-LC Syrup
Each 5 ml contains :
Montelukast Sodium IP
eq. to Montelukast 4 mg
Levocetirizine Hydrochloride IP 2.5 mg
In a flavoured syrupy base q.s.
-
Quick View
MONET-LC Tablets
Each film coated tablet contains:
Montelukast Sodium IP
eq. to Montelukast 10 mg
Levocetirizine Hydrochloride IP 5 mg
-
Quick View
-
Quick View
Maprazyme Liquid
Each 5 ml. contains:
Diastase (1:1200) IP 50 mg
(Fungal Diastase derived from
Aspergillus oryzae – Digests not
less than 60 gm. of cooked starch)
Pepsin (1:3000) IP 10 mg
(Digests not less than 30 gm. of
coagulated egg albumin)
-
Quick View
-
Quick View
Rabetac-D Capsules
Each hard gelatin capsule contains:
Rabeprazole Sodium IP 20 mg
(As enteric coated pellets)
Domperidone IP 30 mg
(As sustained release pellets)
-
Quick View
-
Quick View
LAXIL 200
Each 5 ml. contains:
Lactulose Concentrate USP
eq. to lactulose 3.35 gm
in a palatable base : q.s.
-
Quick View
BESTALAC Sachet
Each 1 g Sachet contains:
Lactobacillus acidophilus 0.24 billion
Bifidobacterium longum 0.24 billion
Streptococcus thermophilus 0.24 billion
Saccharomyces boulardii 0.05 billion
Fructo Oligo Saccharides 300 mg
-
Quick View
BESTALAC Capsules
Each hard gelatin capsule contains:
Lactobacillus rhamnosus 0.48 billion
Bifidobacterium longum 0.48 billion
Streptococcus thermophilus 0.48 billion
Saccharomyces boulardii 0.10 billion
Fructo Oligo Saccharides 300 mg
-
Quick View
Ageflo 200
Each 5 ml contains :
Potassium Citrate Monohydrate IP 1100 mg.
Citric Acid Monohydrate IP 334 mg.
Flavoured Sorbitol Base q.s.
(Each ml contains 2mEq of Potassium ion)
-
Quick View
-
Quick View
Coaid Syrup
Each 5 ml (one teaspoonful) contains:
Simethicone Emulsion USP
eq. to Simethicone USP 40 mg.
Dill Oil BP 0.025 ml.
Fennel Oil USP 0.0035 ml.
Syrupy base q.s.
-
Quick View
Coaid Drops
Each ml. (approx. 20 drops) contains :
Simethicone Emulsion USP
equivalent to Simethicone USP 40 mg
Dill Oil BP 0.005 ml
Fennel Oil USP 0.0007 ml
Sucrose base : q.s.
-
Quick View
-
Quick View
-
Quick View
Din-M Tablets
Each film coated tablet contains :
Drotaverine Hydrochloride IP 80 mg
Mefenamic Acid IP 250 mg
-
Quick View
Din-A Tablets
Each film-coated tablet contains :
Drotaverine Hydrochloride IP 80 mg
Aceclofenac IP 100 mg
-
Quick View
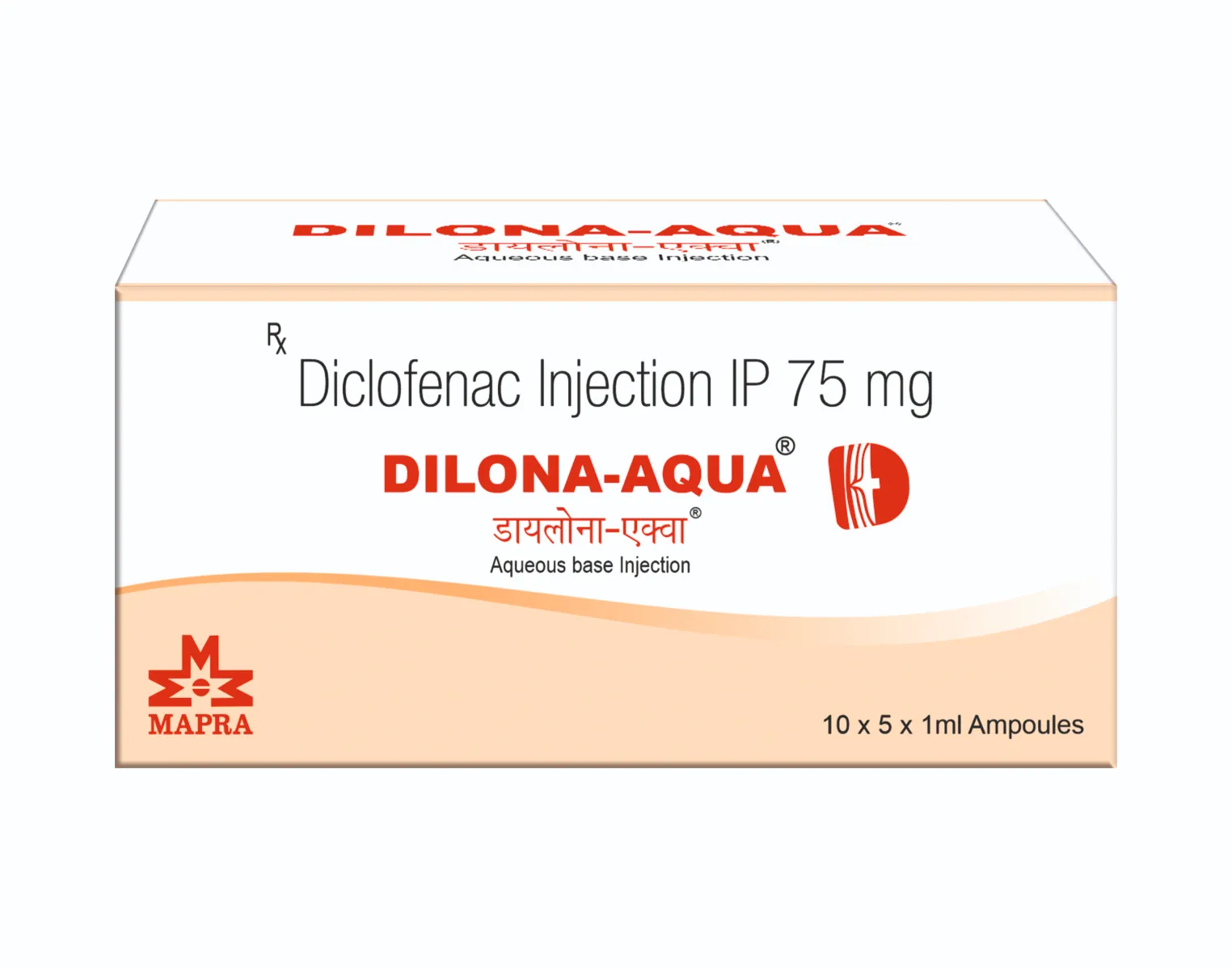
DILONA SPRAY
Diclofenac Diethylamine IP 1.16% w/w
(Equil. to Diclofenac Sodium IP 1.00%)
Linseed Oil BP 3.00% w/w
Methyl Salicylate IP 10.00% w/w
Menthol IP 5.00% w/w
Excipients & Propellant q.s. to 100.00% w/w
-
Quick View
-
Quick View
DILONA GEL
Diclofenac Diethylamine IP 1.16% w/w
(Eq. to Diclofenac Sodium 1.00%w/w)
Oleum Lini
(Containing predominantly alpha-Linolenic acid) 1.00% w/w
Methyl Salicylate IP 10.00% w/w
Menthol IP 5.00% w/w
Benzyl Alcohol IP 1.00% w/w
(as preservative)
Capsaicin USP 0.025% w/w
Gel base q.s.
-
Quick View
-
Quick View
-
Quick View
Dilona-SP Tablets
Each enteric coated tablet contains :
Serratiopeptidase IP (20000 units) 10 mg
Diclofenac Potassium BP 50 mg
-
Quick View
-
Quick View
MAPEPTOZ 1.125GM INJECTION
Each vial contains :
Piperacillin Sodium IP
equivalent to Piperacillin 1 g
Tazobactam Sodium IP
equivalent to Tazobactam 0.125 g
-
Quick View
MAPEPTOZ 2.25 GM INJECTION
Each vial contains :
Piperacillin Sodium IP
equivalent to Piperacillin 2 g
Tazobactam Sodium IP
equivalent to Tazobactam 0.25 g
-
Quick View
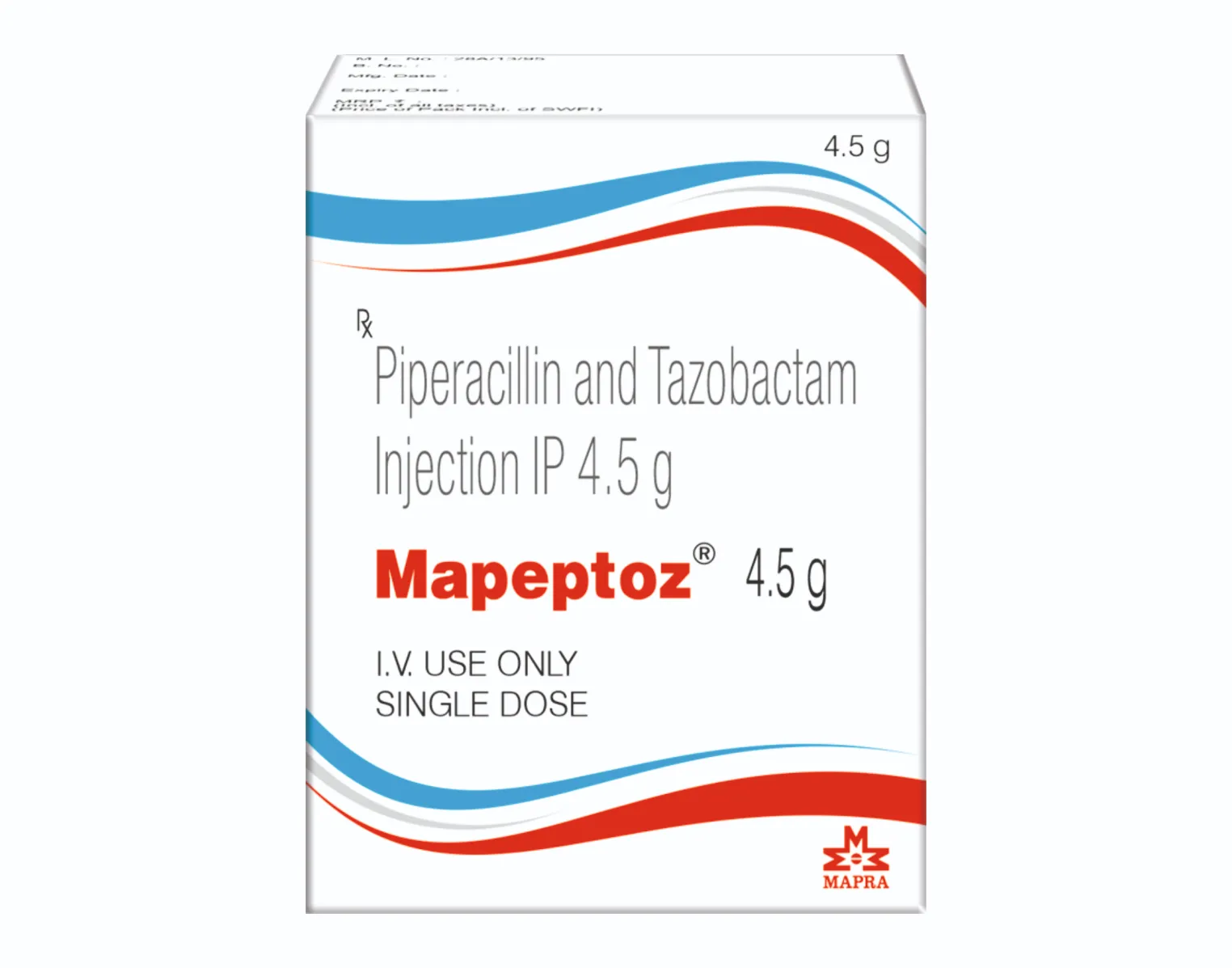
MAPEPTOZ 4.5 GM INJECTION
Each vial contains :
Piperacillin Sodium IP
equivalent to Piperacillin 4 g
Tazobactam Sodium IP
equivalent to Tazobactam 0.5 g
-
Quick View
MEGAZID-XP 281.25 MG INJECTION
Each vial contains:
Ceftazidime Pentahydrate IP
equivalent to Ceftazidime 250 mg
(added Sterile Sodium
Carbonate as buffering agent)
Tazobactam Sodium IP
equivalent to Tazobactam 31.25 mg
-
Quick View
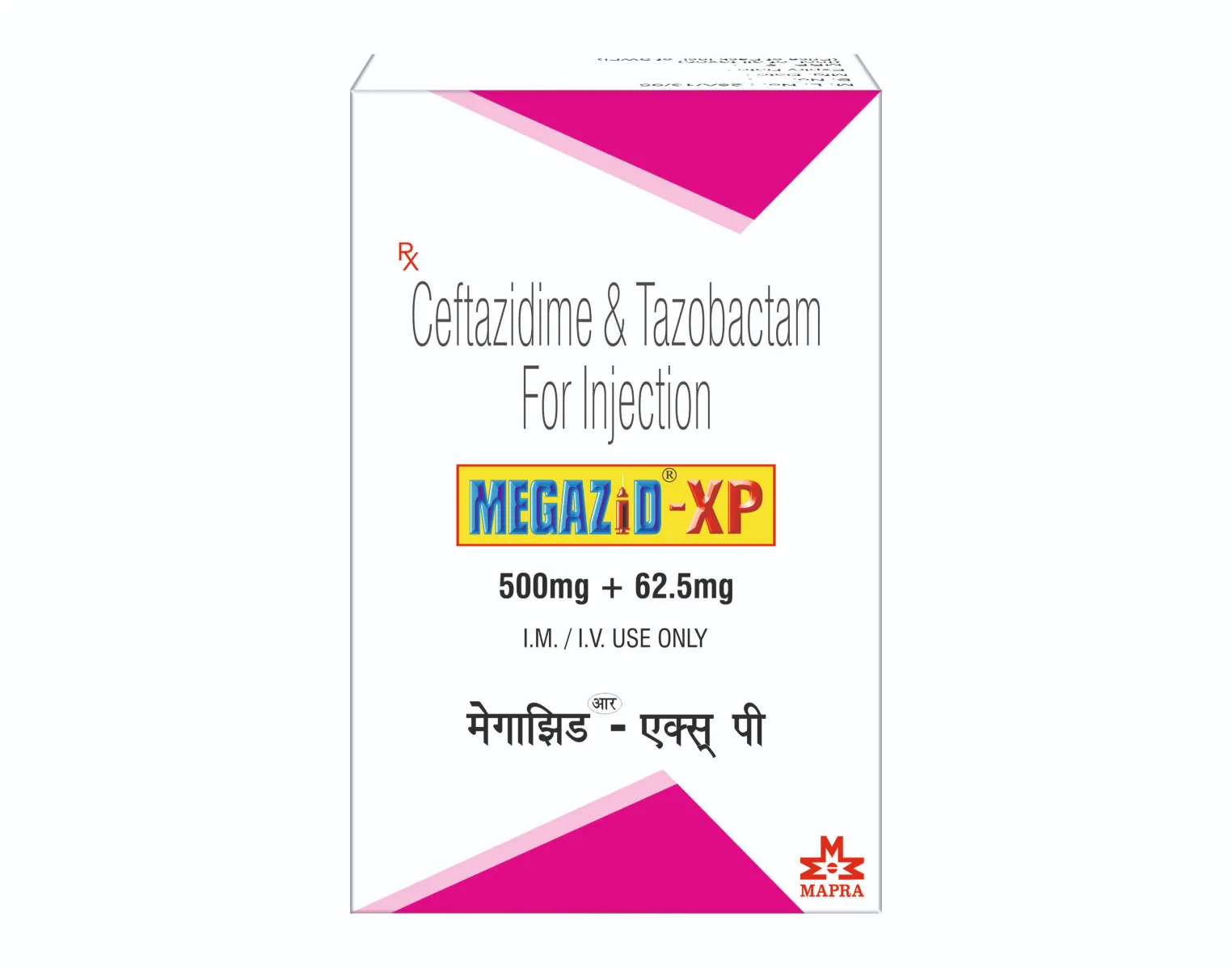
MEGAZID-XP 562.5 MG INJECTION
Each vial contains:
Ceftazidime Pentahydrate IP
equivalent to Ceftazidime 500 mg
(added Sterile Sodium
Carbonate as buffering agent)
Tazobactam Sodium IP
equivalent to Tazobactam 62.5 mg
-
Quick View
MEGAZID-XP 1.125 GM INJECTION
Each vial contains:
Ceftazidime Pentahydrate IP
equivalent to Ceftazidime 1 g
(added Sterile Sodium
Carbonate as buffering agent)
Tazobactam Sodium IP
equivalent to Tazobactam 125 mg
-
Quick View
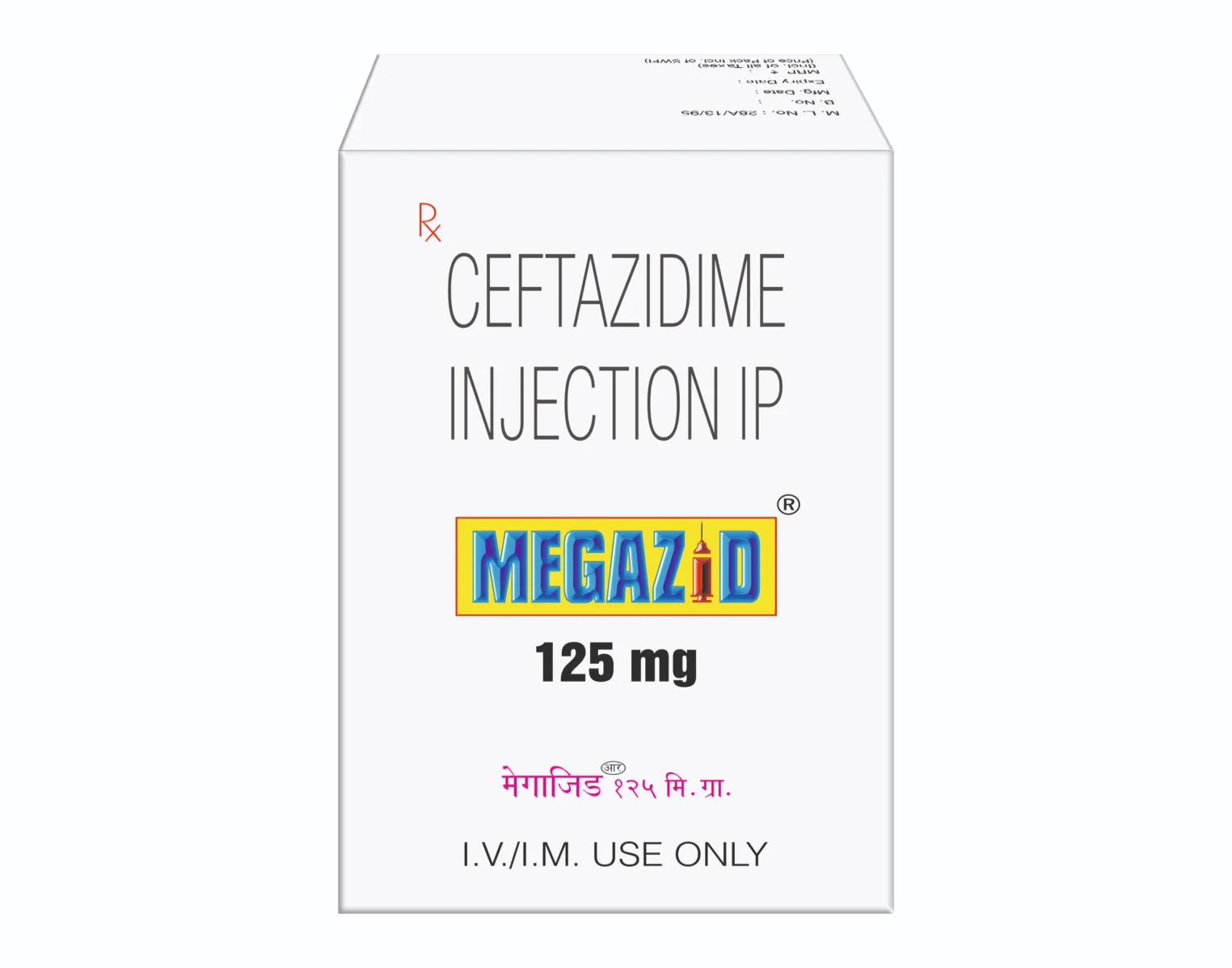
MEGAZID 125 MG INJECTION
Each vial contains:
Ceftazidime for Injection IP
equivalent to Anhydrous Ceftazidime 125 mg
(Ceftazidime for injection is a Sterile Mixture
of Sterile Ceftazidime and Sodium Carbonate)
-
Quick View
MEGAZID 250 MG INJECTION
Each vial contains:
Ceftazidime for Injection IP
equivalent to Anhydrous Ceftazidime 250 mg
(Ceftazidime for injection is a Sterile Mixture
of Sterile Ceftazidime and Sodium Carbonate)
-
Quick View
MEGAZID 1 GM INJECTION
Each vial contains:
Ceftazidime for Injection IP
equivalent to Anhydrous Ceftazidime 1 g
(Ceftazidime for injection is a Sterile Mixture
of Sterile Ceftazidime and Sodium Carbonate)
-
Quick View
TAXONE-XP 250 MG INJECTION
Each vial contains :
Ceftriaxone Sodium IP
equivalent to Ceftriaxone 250 mg
Tazobactam Sodium IP
equivalent to Tazobactam 31.25 mg
-
Quick View
TAXONE-XP 500 MG INJECTION
Each vial contains :
Ceftriaxone Sodium IP
equivalent to Ceftriaxone 500 mg
Tazobactam Sodium IP
equivalent to Tazobactam 62.5 mg
-
Quick View
TAXONE-XP 1 GM INJECTION
Each vial contains :
Ceftriaxone Sodium IP
equivalent to Ceftriaxone 1 g
Tazobactam Sodium IP
equivalent to Tazobactam 125 mg
-
Quick View
-
Quick View
-
Quick View
TAXONE 1 GM INJECTION
Each Film Coated tablet Contains :
Faropenem Sodium Hydrate JP
equivalent to Faropenem 200 mg
-
Quick View
-
Quick View
Suplopen 200 Tablets
Each Film Coated tablet Contains :
Faropenem Sodium Hydrate JP
equivalent to Faropenem 200 mg
-
Quick View
Mapdox 50 Dry Syrup
Each film coated tablet contains :
Cefpodoxime Proxetil IP
equivalent to Cefpodoxime 200 mg
Potassium Clavulanate Diluted IP
equivalent to Clavulanic Acid 125 mg
-
Quick View
Mapdox CV 100 mg Dry Syrup
Each 5 ml of the reconstituted suspension contains:
Cefpodoxime Proxetil IP
equivalent to Cefpodoxime 100 mg
Potassium Clavulanate diluted IP
equivalent to Clavulanic Acid 62.5 mg
-
Quick View
Mapdox CV 200 mg Tablets
Each film coated tablet contains :
Cefpodoxime Proxetil IP
equivalent to Cefpodoxime 200 mg
Potassium Clavulanate Diluted IP
equivalent to Clavulanic Acid 125 mg
-
Quick View
-
Quick View
Taxone-OF Tabs
Each film-coated tablet contains :
Cefixime IP as Trihydrate
equivalent to Anhydrous Cefixime 200 mg
Ofloxacin IP 200 mg
-
Quick View
TAXONE-O Drops
Each ml of reconstituted suspension contains:
Cefixime IP as Trihydrate
equivalent to Anhydrous Cefixime 25 mg
-
Quick View
TAXONE-O Dry Syrup
Each combipack contains :
(A) 1 Vial of Amoxycillin and Potassium Clavulanate
Oral Suspension IP
Each 5 ml of Reconstituted
suspension contains :
Amoxycillin Trihydrate IP
Potassium Clavulanate Diluted IP
(B) 1 Vial of Sterile Water for Reconstitution 30ml
-
Quick View
TAXONE-O DT 200 mg Tablets
Each uncoated dispersible tablet contains:
Cefixime IP as Trihydrate
equivalent to Anhydrous Cefixime 100 mg
-
Quick View
TAXONE-O DT 100 mg Tablets
Each uncoated dispersible tablet contains:
Cefixime IP as Trihydrate
equivalent to Anhydrous Cefixime 100 mg
-
Quick View
TRIMOX-CV 228.5 Dry Syrup
Each 5 ml of the reconstituted suspension contains :
Amoxycillin Trihydrate IP
equivalent to Amoxycillin 200 mg
Potassium Clavulanate Diluted IP
equivalent to Clavulanic Acid 28.5 mg
-
Quick View
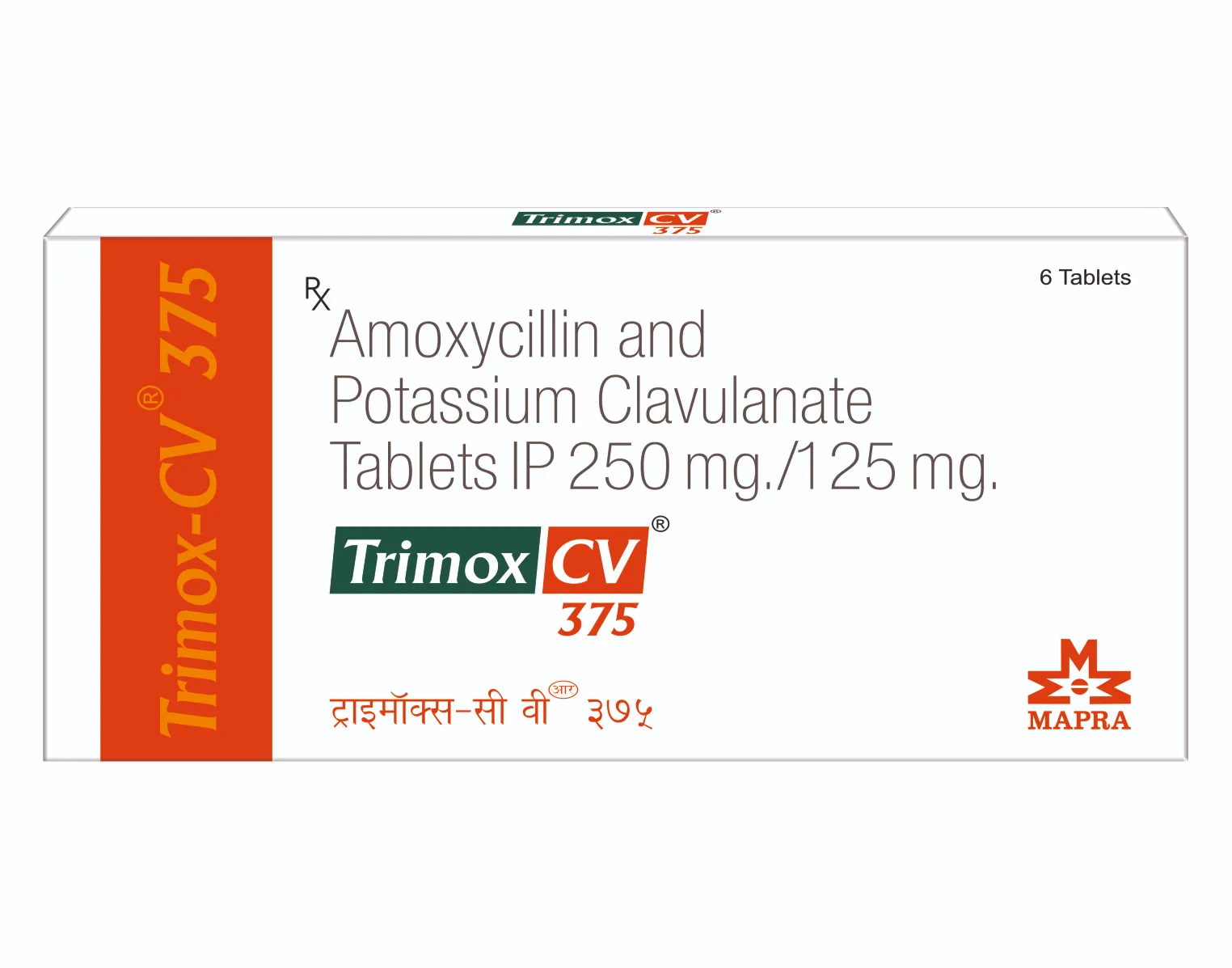
TRIMOX-CV 375 mg Tablets
Each Film-coated tablet contains :
Amoxycillin Trihydrate IP
equivalent to Amoxycillin 250 mg
Potassium Clavulanate Diluted IP
equivalent to Clavulanic Acid 125 mg
-
Quick View
TRIMOX-CV 625 mg Tablets
Each Film-coated tablet contains :
- Amoxycillin Trihydrate IP
equivalent to Amoxycillin 500 mg
- Potassium Clavulanate Diluted IP
equivalent to Clavulanic Acid 125 mg
-
Quick View
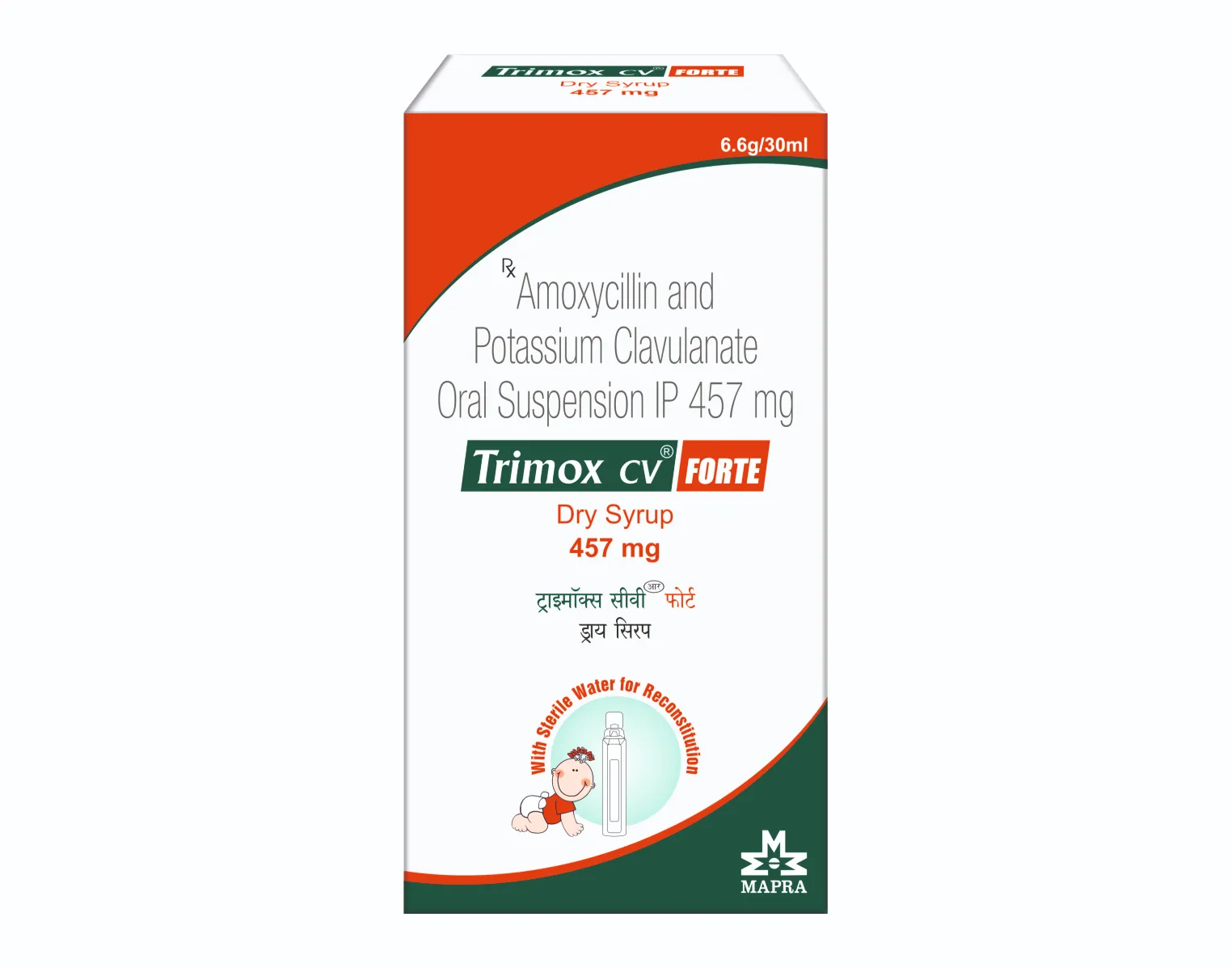
TRIMOX-CV FORTE Dry Syrup
Each combi pack contains :
(A) 1 Vial of Amoxycillin and Potassium Clavulanate
Oral Suspension IP
Each 5 ml of Reconstituted suspension contains :
Amoxycillin Trihydrate IP
equivalent to Amoxycillin 400 mg
Potassium Clavulanate Diluted IP
equivalent to Clavulanic Acid 57 mg
(B) 1 Vial of Sterile Water for Reconstitution 30 ml